RESEARCH
Past Research
Membrane Permeation
Membranes play a myriad of essential roles in biology, acting not only as the primary means of compartmentalization, but also mediating and influencing communication between those compartments. Atomistic simulations are a powerful way to learn about membranes. Our group is pushing the limits of atomistic simulations to incorporate the heterogeneity of realistic membranes and understand their interactions with amphiphiles, toxins, charged small molecules, and membrane proteins.
Mycolactone, a toxin produced by Mycobacterium ulcerans, causes the neglected tropical skin disease Buruli ulcer by inhibiting peptide translocation through the Sec61 translocon. Only one of two isoforms (B) is cytotoxic. This work probes the roles of cellular localization versus association in isomer specificity. Simulations reveal that stronger membrane affinity increases the local concentration of isomer B. Concomitantly, mycolactone B interacts more potently with the translocon through patch residues involved in signal recognition during co-translation and through gating elements during post-translational translocation. Thus, isomer specificity is a consequence of both localization and association revealing molecular features that may benefit the development of Buruli ulcer diagnostics and Sec61 therapeutics.
Cover story
Capturing the Liquid-crystalline Phase Transformation:
Implications for Protein Targeting to Sterol Ester-rich Lipid Droplets
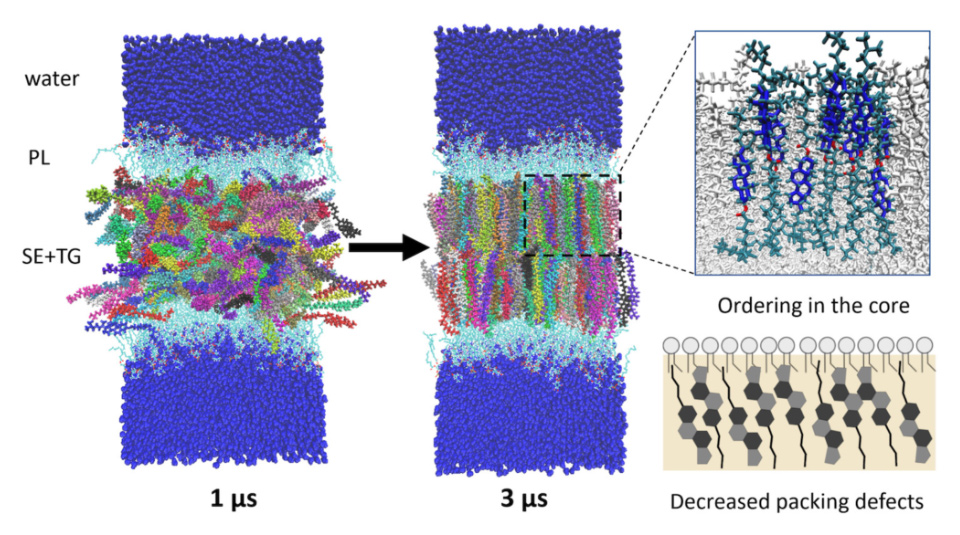
Lipid droplets (LDs), hubs of neutral lipid storage and distribution, have been shown in cryoEM to undergo a liquid-crystalline phase transition under high sterol ester (SE) concentrations, forming concentric rings like layers of an onion. This research sought to characterize this phase transformation at the molecular level. After developing sterol ester parameters, we were able to capture this phase transformation with 90% SE in 3 microseconds of simulation. Interestingly, the ordered layers of SEs in the LD core transformed the phospholipid monolayer surrounding the LD as well: increasing lipid packing and tail order parameters, while squeezing out surface neutral lipids and significantly diminishing membrane packing defects. We discuss how these changes will have profound effects on LD-protein interactions, consistent with experimentally demonstrated altered LD proteomes.